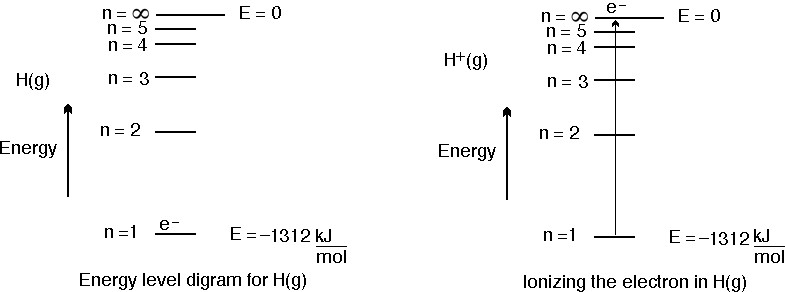
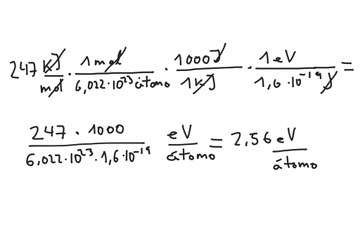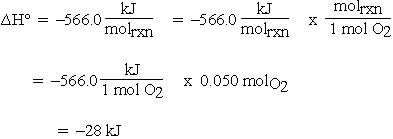How much energy in KJ/mol is released when an electron makes a transition from n=5 to n=2 in a hydrogen atom? | Homework.Study.com
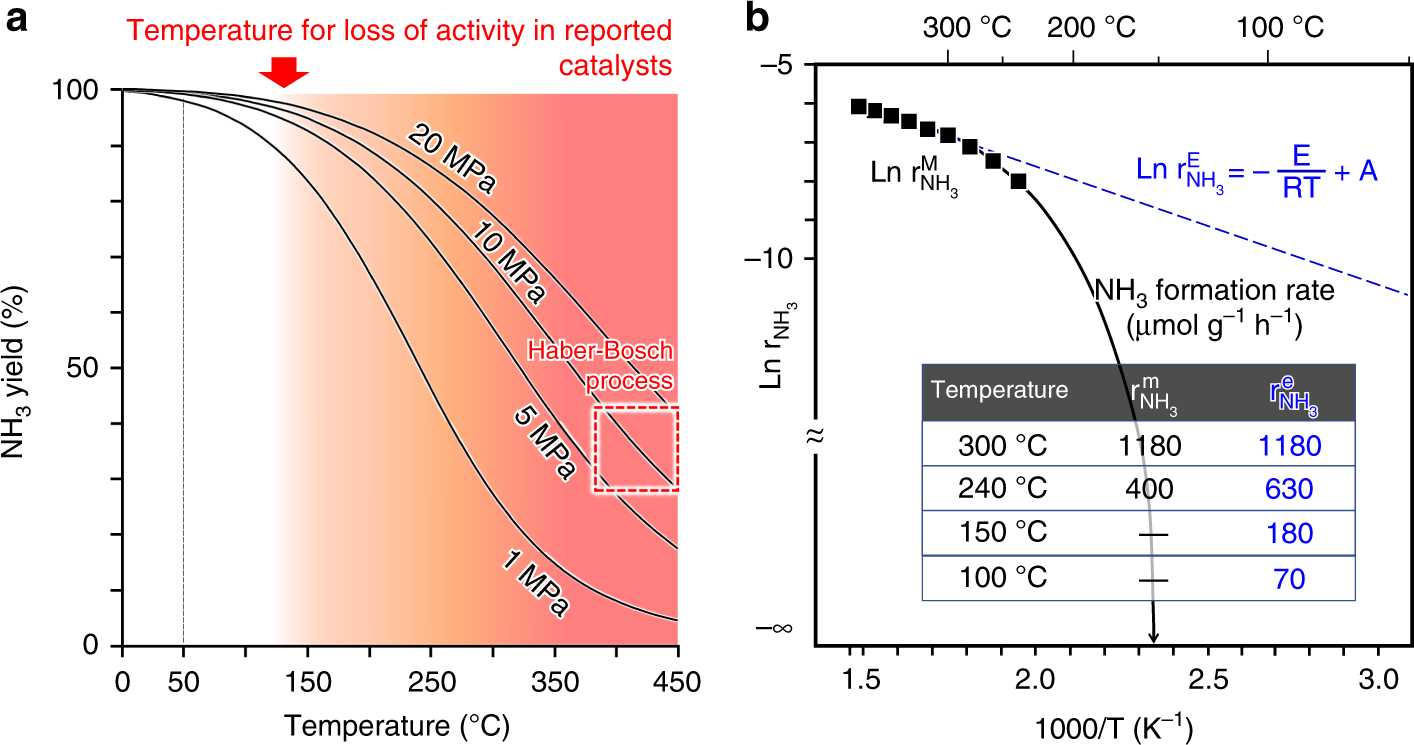
Solid solution for catalytic ammonia synthesis from nitrogen and hydrogen gases at 50 °C | Nature Communications
74 A hydrogen like system has ionization energy 11808kJ/mol. Find number of proton in nucleus of that system.